 W is the net work done by the system-that is, W is the sum of all work done on or by the system. Q is the net heat transferred into the system-that is, Q is the sum of all heat transfer into and out of the system. 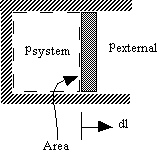
Here Δ U is the change in internal energy U of the system. In equation form, the first law of thermodynamics is Δ U = Q − W. The first law of thermodynamics states that the change in internal energy of a system equals the net heat transfer into the system minus the net work done by the system. The first law of thermodynamics applies the conservation of energy principle to systems where heat transfer and doing work are the methods of transferring energy into and out of the system. If we are interested in how heat transfer is converted into doing work, then the conservation of energy principle is important. As the entire system gets hotter, work is done-from the evaporation of the water to the whistling of the kettle. 
The water in the kettle is turning to water vapor because heat is being transferred from the stove to the kettle. This boiling tea kettle represents energy in motion.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
